Meningitis Vaccine Project (MVP)
"There is no point in having a vaccine if you cannot roll it out. This is a story for donors, who increasingly need to know that the money which is going into developing countries is actually having an impact, and this is a very good example of something which has a clear and measurable impact." - Helen Evans, interim chief executive officer, GAVI, Kyodo News, December 7 2010.
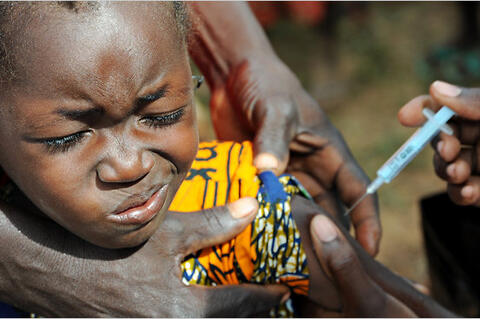
The Meningitis Vaccine Project (MVP) was created in 2001 as a partnership between PATH and the World Health Organization (WHO) with the goal of eliminating meningitis as a public health problem in sub-Saharan Africa through the development, testing, introduction, and widespread use of conjugate meningococcal vaccines. Among its objectives has been the introduction of the vaccine for group A meningitis, MenAfriVac® (Serum Institute of India Ltd.), through mass and routine immunisation in synergy with other public health programmes.
The vaccine, intended for people from ages 1 to 29, was developed through "facilitating and coordinating numerous partnerships across the world....This approach has... the potential for providing a new model for vaccine development that could facilitate the introduction of other 'orphan' vaccines whose primary markets are low-income countries in the developing world." The vaccine became licensed and WHO-pre-qualified for use in 2010.
MVP's strategy has been based on the following set of principles:
- "The project aims to achieve a sustainable public health impact and is not simply about making vaccines available.
- Decisions about vaccine development are linked to introduction strategies and likely financial constraints.
- African public health officials and other stakeholders in Africa are closely involved with MVP. In addition, MVP emphasizes the need for understanding country needs and perspectives."
Following the strategy of an initial mass vaccination programme and then an integration of the vaccine into routine childhood immunisation programmes, the vaccine was introduced at large scale in meningitis-belt countries in 2010. "Since then, more than 217 million people in 15 countries have received the vaccine."
According to Dr. Kathy Neuzil, director of Vaccine Access and Delivery at PATH, "Step one involved listening to the affected countries. Health ministers wanted a practical, affordable vaccine to alleviate the suffering of their people.
Step two focused on developing the tactics and strategy to achieve the goal. The latter wasn't easy, and as one door closed, the MVP team made sure to open others, even when they were stuck, barred shut, or seemingly too small. If there was no door, then the team looked for a window."
Trough work among global partners, the vaccine received approval for infants. Country partners, involved from the project's inception, worked on ensuring a vaccine price, supply, and implementation plan that focused on sustainability, such that the project has closed as of December 31 2014 and left routine immusation in the hands of its country partners, either incorporating it in Expanded Programme on Immunization (EPI) or organising campaigns for immunisation of children aged 1 to 4 years of age every 5 years: "Ethiopia completes the second phase of the MenAfriVac campaign (phase 3 scheduled in 2015), Nigeria completes its fourth (and last) vaccination campaign. Côte d'Ivoire, Mauritania, and Togo introduce MenAfriVac."
Prior to closing, MVP developed a post introduction evaluation plan to assess the impact of the introduction of MenAfriVac® in the first introduction countries: "Monitoring at country level will be carried out by country-based surveillance units with technical support from WHO, UNICEF [the United Nations Children's Fund], and other partners, including the London School of Hygiene and Tropical Medicine, who is in charge of conducting a series of carriage studies to evaluate the impact of MenAfriVac® on the transmission of meningococcal infection in countries of the African meningitis belt."
Children, Immunisation and Vaccination
"Following the devastating epidemic of 1996-1997 (more than 250,000 cases of disease and over 25,000 deaths), African leaders called for the development of a vaccine that would eliminate, once and for all, group A meningitis epidemics in Africa."
WHO, PATH, Serum Institute of India Ltd. (SIIL), Synco Bio Partners B.V., United States Food and Drug Administration, United Kingdom National Institute for Biological Standards and Control (NIBSC), the International Coordinating Group (ICG) on Vaccine Provision for Epidemic Meningitis Control, Agence de Médecine Préventive (AMP), Médecins Sans Frontières (MSF), and UNICEF. Click here for more partners’ information.
Email from Claire Hudson to The Communication Initiative on January 26 2015 and the MVP website, January 28 2015. Image credit: Barry Rodrigue/World Health Organization
- Log in to post comments












































